FIELD: chemistry.
SUBSTANCE: in new compounds with formula (I): 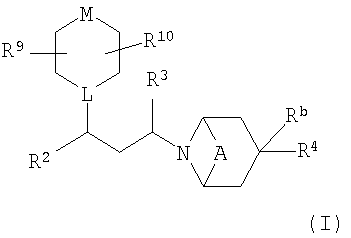 (I) A is absent or represents (CH2)2; L is CH or N; M is NR1, O, S, S(O) or S(O)2; R1 is C1-6alkyl, substituted with phenyl {which itself is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy, CF3}; phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy, CF3, C1-4alkylthio}, S(O)2R, S(O)2NR6R7, C(O)R8; R2 is phenyl (which is possibly substituted with halogen, CN or C1-4halogenalkyl), thienyl or halogenthienyl; R3 is hydrogen or methyl; Rb is hydrogen or C1-3alkyl; R4 is a five- or six-member heterocycle, containing at least one carbon atom, one to four nitrogen atoms and, possibly, one oxygen or sulphur atom, where the carbon atom in the said heterocycle R4 is possibly substituted with oxo, C1-6alkyl [which is possibly substituted with halogen, OH, C1-4alkoxy, S(C1-4alkyl) group or piperidinyl {which it self is possibly substituted with benzene [which is possibly substituted with a S(O)2(C1-4alkyl) group], C(O)(C1-4alkoxy) group, C(O)NH2, C(O)NH(C1-4alkyl), C(O)N(C1-4alkyl)2 or S(O)2(C1-4alkyl) [where alkyl is possibly substituted with fluoro]}], C3-6cycloalkyl, CN, C(O)NH2, C(O)NH(phenylC1-2alkyl) group, phenyl [which is possibly substituted with a S(O)2(C1-4alkyl) group] or benzyl [which is possibly substituted with a S(O)2(C1-4alkyl) group]; if possible, the nitrogen atom in the said heterocycle R4 is substituted with C1-6alkyl [which is possibly substituted with C1-4alkoxy, S(O)(C1-4alkyl) group, S(O)2(C1-4alkyl), C(O)(C1-4alkoxy), CONH2, CONH(C1-4alkyl), CON(C1-4alkyl)2, phenyl{which is possibly substituted with C1-4alkyl, C1-4alkoxy, S(O)(C1-4alkyl) group or S(O)2(C1-4alkyl)}, piperidinyl {which is possibly substituted with a S(O)(C1-4alkyl) group or S(O)2(C1-4alkyl)}], C3-6cycloalkyl, CO(C1-4alkyl) group [which is possibly substituted with a halogen], S(O)2(C1-4alkyl) group [which is possibly substituted with fluorine], COO(C1-6alkyl) group, phenyl [which is possibly substituted with a S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) group]; - under the condition that the nitrogen atom in the said heterocycle R4 is substituted with an alky group, the said alkyl does not have C1-4alkoxy, S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) substitute on the carbon atom, bonded to the nitrogen atom in the said heterocycle R4; - five- or six-member heterocyle R4 is possibly condensed with cyclohexane, piperadine, benzole, pyridine, pyridazine, pyrimidine or pyrazine ring; ring carbon atoms in the said condensed cyclohexane, piperadine, benzole, pyridine, pyridazine, pyrimidine or pyrazine ring are possibly substituted with a halogen, C1-4alkyl, C1-4alkoxy, CF3, S(C1-4alkyl), S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) group; and the nitrogen atom of the condensed piperidine ring is possibly substituted with C1-4alkyl [which is possibly substituted with oxo, halogen, OH, C1-4alkoxy, C(O)(C1-4alkoxy), C(O)NH2, C(O)NH(C1-4alkyl) group, C(O)N(C1-4alkyl)2 group, C(O)(C1-4alkyl)group [where alkyl is possibly substituted with C1-4alkoxy or halogen], benzene [which is possibly substituted with S(O)(C1-4alkyl) or S(O)2(C1-4alkyl)], C(O)(C1-4alkoxy), C(O)NH2, C(O)NH(C1-4alkyl), C(O)N(C1-4alkyl)2 or S(O)2(C1-4alkyl) group [where alkyl is possibly substituted with fluoro]; R5 is C1-6alkyl [which is possibly substituted with a halogen (for example fluoro), C1-4alkoxy, phenyl {which itself is possibly substituted with a halogen, C1-4alkyl, C1-4alkoxy}], C3-7cycloalkyl (which is possibly substituted with a halogen or C1-6alkyl), piranyl, phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}, or a 5- or 6-member saturated nitrogen-containing heterocyclic ring {which is possibly substituted with a S(O)2(C1-4alkyl) or C(O)(C1-4alkyl) group}; R8 is hydrogen, C1-4alkyl [which is possibly substituted with halogen (for example fluro), C1-4alkoxy, phenyl{which itself is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}], C3-7cycloalkyl (which is possibly substituted with halogen or C1-4alkyl), piranyl, phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}, or a 5- or 6-member saturated nitrogen-containing heterocyclic ring {which is possibly substituted with S(O)2(C1-4alkyl) or C(O)(C1-4alkyl) group}; R6 and R7 are bonded, forming a 5- or 6-member ring which is possibly substituted with C1-4alkyl; R9 and R10 independently represent hydrogen or C1-6alkyl; or to its pharmaceutically acceptable salts. The invention also relates to a method of obtaining compounds in paragraph 1, to a method of modulating activity of CCR5 receptor, as well as to a pharmaceutical composition.
(I) A is absent or represents (CH2)2; L is CH or N; M is NR1, O, S, S(O) or S(O)2; R1 is C1-6alkyl, substituted with phenyl {which itself is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy, CF3}; phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy, CF3, C1-4alkylthio}, S(O)2R, S(O)2NR6R7, C(O)R8; R2 is phenyl (which is possibly substituted with halogen, CN or C1-4halogenalkyl), thienyl or halogenthienyl; R3 is hydrogen or methyl; Rb is hydrogen or C1-3alkyl; R4 is a five- or six-member heterocycle, containing at least one carbon atom, one to four nitrogen atoms and, possibly, one oxygen or sulphur atom, where the carbon atom in the said heterocycle R4 is possibly substituted with oxo, C1-6alkyl [which is possibly substituted with halogen, OH, C1-4alkoxy, S(C1-4alkyl) group or piperidinyl {which it self is possibly substituted with benzene [which is possibly substituted with a S(O)2(C1-4alkyl) group], C(O)(C1-4alkoxy) group, C(O)NH2, C(O)NH(C1-4alkyl), C(O)N(C1-4alkyl)2 or S(O)2(C1-4alkyl) [where alkyl is possibly substituted with fluoro]}], C3-6cycloalkyl, CN, C(O)NH2, C(O)NH(phenylC1-2alkyl) group, phenyl [which is possibly substituted with a S(O)2(C1-4alkyl) group] or benzyl [which is possibly substituted with a S(O)2(C1-4alkyl) group]; if possible, the nitrogen atom in the said heterocycle R4 is substituted with C1-6alkyl [which is possibly substituted with C1-4alkoxy, S(O)(C1-4alkyl) group, S(O)2(C1-4alkyl), C(O)(C1-4alkoxy), CONH2, CONH(C1-4alkyl), CON(C1-4alkyl)2, phenyl{which is possibly substituted with C1-4alkyl, C1-4alkoxy, S(O)(C1-4alkyl) group or S(O)2(C1-4alkyl)}, piperidinyl {which is possibly substituted with a S(O)(C1-4alkyl) group or S(O)2(C1-4alkyl)}], C3-6cycloalkyl, CO(C1-4alkyl) group [which is possibly substituted with a halogen], S(O)2(C1-4alkyl) group [which is possibly substituted with fluorine], COO(C1-6alkyl) group, phenyl [which is possibly substituted with a S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) group]; - under the condition that the nitrogen atom in the said heterocycle R4 is substituted with an alky group, the said alkyl does not have C1-4alkoxy, S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) substitute on the carbon atom, bonded to the nitrogen atom in the said heterocycle R4; - five- or six-member heterocyle R4 is possibly condensed with cyclohexane, piperadine, benzole, pyridine, pyridazine, pyrimidine or pyrazine ring; ring carbon atoms in the said condensed cyclohexane, piperadine, benzole, pyridine, pyridazine, pyrimidine or pyrazine ring are possibly substituted with a halogen, C1-4alkyl, C1-4alkoxy, CF3, S(C1-4alkyl), S(O)(C1-4alkyl) or S(O)2(C1-4alkyl) group; and the nitrogen atom of the condensed piperidine ring is possibly substituted with C1-4alkyl [which is possibly substituted with oxo, halogen, OH, C1-4alkoxy, C(O)(C1-4alkoxy), C(O)NH2, C(O)NH(C1-4alkyl) group, C(O)N(C1-4alkyl)2 group, C(O)(C1-4alkyl)group [where alkyl is possibly substituted with C1-4alkoxy or halogen], benzene [which is possibly substituted with S(O)(C1-4alkyl) or S(O)2(C1-4alkyl)], C(O)(C1-4alkoxy), C(O)NH2, C(O)NH(C1-4alkyl), C(O)N(C1-4alkyl)2 or S(O)2(C1-4alkyl) group [where alkyl is possibly substituted with fluoro]; R5 is C1-6alkyl [which is possibly substituted with a halogen (for example fluoro), C1-4alkoxy, phenyl {which itself is possibly substituted with a halogen, C1-4alkyl, C1-4alkoxy}], C3-7cycloalkyl (which is possibly substituted with a halogen or C1-6alkyl), piranyl, phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}, or a 5- or 6-member saturated nitrogen-containing heterocyclic ring {which is possibly substituted with a S(O)2(C1-4alkyl) or C(O)(C1-4alkyl) group}; R8 is hydrogen, C1-4alkyl [which is possibly substituted with halogen (for example fluro), C1-4alkoxy, phenyl{which itself is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}], C3-7cycloalkyl (which is possibly substituted with halogen or C1-4alkyl), piranyl, phenyl {which is possibly substituted with halogen, C1-4alkyl, C1-4alkoxy}, or a 5- or 6-member saturated nitrogen-containing heterocyclic ring {which is possibly substituted with S(O)2(C1-4alkyl) or C(O)(C1-4alkyl) group}; R6 and R7 are bonded, forming a 5- or 6-member ring which is possibly substituted with C1-4alkyl; R9 and R10 independently represent hydrogen or C1-6alkyl; or to its pharmaceutically acceptable salts. The invention also relates to a method of obtaining compounds in paragraph 1, to a method of modulating activity of CCR5 receptor, as well as to a pharmaceutical composition.
EFFECT: obtaining new biologically active compounds with modulating effect towards CCR5 receptor.
15 cl, 29 ex, 12 tbl
| Title | Year | Author | Number |
|---|---|---|---|
| PYRROLOTRIAZINE ANILINE DERIVATIVES EFFECTIVE AS KINASE INHIBITORS | 2003 |
|
RU2375363C2 |
| PIPERIDINE DERIVATIVES AS MODULATORS OF ACTIVITY OF CCR5 RECEPTOR, METHOD AND INTERMEDIATE COMPOUNDS FOR OBTAINING THEM, AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | 2002 |
|
RU2345990C2 |
| USING SIP RECEPTOR MODULATOR | 2008 |
|
RU2498796C2 |
| SUBSTITUTED QUINAZINE DERIVATIVES AS FGFR-KINASE INHIBITORS FOR TREATMENT OF CANCER | 2011 |
|
RU2602233C2 |
| QUINAZOLINONE DERIVATIVES APPLICABLE AS MODULATORS OF FGFR KINASE | 2014 |
|
RU2701517C2 |
| NEW PIPERIDINES AS CHEMOKINE (CCR) MODULATORS | 2005 |
|
RU2348616C2 |
| NEW 5,6-DIHYDROPYRIDINE-2-ON COMPOUNDS APPLICABLE AS THROMBIN INHIBITORS | 2004 |
|
RU2335492C2 |
| QUINOLINES AS FGFR KINASE MODULATORS | 2012 |
|
RU2625303C2 |
| PTERIDINES AS FGFR INHIBITORS | 2013 |
|
RU2702906C2 |
| OPTIONALLY CONDENSED HETEROCYCLYL-SUBSTITUTED PYRIMIDINE DERIVATIVES SUITABLE FOR TREATING INFLAMMATORY, METABOLIC, ONCOLOGICAL AND AUTOIMMUNE DISEASES | 2015 |
|
RU2719422C2 |
Authors
Dates
2009-09-27—Published
2005-04-20—Filed