FIELD: peptide chemistry. SUBSTANCE: product: derivatives of peptideamides of the formula (I) (X)n-A-NH2 where X - Ac-L (or D)-(Hal), p-Cl-L (or D)-Phe, L (or D)-Ser, L (or D)-Tyr, L (or D)-Trp, L (or D)-Ser(α-L-Phe), L (or D)-Leu, L (or D)-Arg, L (or D)-Pro, p-Slu, His, D-Ser(t-Bu); n = 3-10; A - azaglycine, or their biologically compatible acetates or hydrochlorides. Reagent 1: compound of the formula (II)  where Y1-Y5 - hydrogen; C1-C4-alkoxy-group; -(CH2)m-C(O)-OH [radicals are similar or different but at least one of their is (CH2)n-C(O)-OH]; m = 1-3; R1 - hydrogen; 4-methoxyphenyl. Reagent 2: silyling agent of the group: tert.-butyldimethylsilyl chloride, tert.-butylphenylsilyl chloride, trimethylchlorosilane, bis-trimethylsilylacetamide. Reaction conditions: process is carried out in the medium of solvent following by treatment of silylated compound with chloroformic acid derivative. Compounds of the formula (III) R2
where Y1-Y5 - hydrogen; C1-C4-alkoxy-group; -(CH2)m-C(O)-OH [radicals are similar or different but at least one of their is (CH2)n-C(O)-OH]; m = 1-3; R1 - hydrogen; 4-methoxyphenyl. Reagent 2: silyling agent of the group: tert.-butyldimethylsilyl chloride, tert.-butylphenylsilyl chloride, trimethylchlorosilane, bis-trimethylsilylacetamide. Reaction conditions: process is carried out in the medium of solvent following by treatment of silylated compound with chloroformic acid derivative. Compounds of the formula (III) R2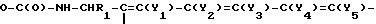 were prepared where Y1-Y5 and R1 - as indicated above; R2 - halogeno (or nitro)-phenyl. These compounds were treated with hydrazide hydrochloride R3-protected with corresponding α-amino acid (X meaning) where R3, Fmoc Bpoc, BOC - are protective groups, in the medium of solvent with formation of compound of the formula (IV)
were prepared where Y1-Y5 and R1 - as indicated above; R2 - halogeno (or nitro)-phenyl. These compounds were treated with hydrazide hydrochloride R3-protected with corresponding α-amino acid (X meaning) where R3, Fmoc Bpoc, BOC - are protective groups, in the medium of solvent with formation of compound of the formula (IV)  . Protective group -BOC- is removed by hydrogenation on the Pt-catalyst. Before the further reaction protective group is conversed to the Fmoc- or BOC-protective group. Then compounds of the formula (IV) (at meaning R3 - Fmoc, Bpoc-urethane-protective group) were copulated using usual peptide reagent (through -(CH)n-C(O)-OH-grouping on the resin), protective group R3 is split, and then step by step Fmoc- or Bpoc-protected α-amino acids (in some cases in the form of activated derivatives) were joined. After synthesis termination of peptide of the formula (I) the latter is released from resin by treatment with moderate force acid. Simultaneously or using conventional procedures temporarily added side chain protective groups were split. EFFECT: improved method of synthesis.
. Protective group -BOC- is removed by hydrogenation on the Pt-catalyst. Before the further reaction protective group is conversed to the Fmoc- or BOC-protective group. Then compounds of the formula (IV) (at meaning R3 - Fmoc, Bpoc-urethane-protective group) were copulated using usual peptide reagent (through -(CH)n-C(O)-OH-grouping on the resin), protective group R3 is split, and then step by step Fmoc- or Bpoc-protected α-amino acids (in some cases in the form of activated derivatives) were joined. After synthesis termination of peptide of the formula (I) the latter is released from resin by treatment with moderate force acid. Simultaneously or using conventional procedures temporarily added side chain protective groups were split. EFFECT: improved method of synthesis.
| Title | Year | Author | Number |
|---|---|---|---|
| PEPTIDES AND THEIR PHYSIOLOGICALLY ACCEPTABLE SALTS | 1992 |
|
RU2083586C1 |
| PEPTIDE DERIVATIVES | 1992 |
|
RU2081880C1 |
| NEUROMEDIN AND SOMATOSTATIN RECEPTOR AGONISTS | 2000 |
|
RU2263680C2 |
| CYCLOHEXAPEPTIDES, MIXTURES THEREOF, AND METHOD OF PREPARING THEREOF | 1995 |
|
RU2163242C2 |
| PTH-COMPOUNDS, METHOD OF THEIR SYNTHESIS, PHARMACEUTICAL COMPOSITION CONTAINING THEIR, DNA FRAGMENT AND FUSED PROTEIN | 1993 |
|
RU2130945C1 |
| ANALOGUES OF PEPTIDE LH-RF, THEIR APPLICATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEREOF | 1998 |
|
RU2212247C2 |
| METHOD OF SOLID-PHASE PEPTIDE SYNTHESIS | 1991 |
|
RU2043362C1 |
| PEPTIDE COMPOSITIONS | 2014 |
|
RU2725150C2 |
| METASTIN DERIVATIVES AND APPLICATION THEREOF | 2006 |
|
RU2430107C2 |
| METASTIN DERIVATIVES AND USE THEREOF | 2007 |
|
RU2454425C2 |
Authors
Dates
1995-05-27—Published
1991-08-29—Filed