FIELD: organic chemistry, medicine, oncology, pharmacy.
SUBSTANCE: invention relates to new derivatives of quinazoline of the formula (I):
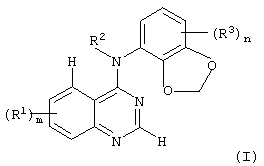
wherein m = 0, 1, 2 or 3; each group R1 that can be similar or different is taken among halogen atom, trifluoromethyl, hydroxy-, amino-group, (C1-C6)-alkyl, (C2-C8)-alkenyl, (C2-C8)-alkynyl, (C1-C6)-alkoxy-, (C2-C6)-alkenyloxy-, (C2-C6)-alkynyloxy-, (C1-C6)-alkylamino-, di-[(C1-C6)-alkyl]amino- and (C2-C6)-alkanoylamino-group, or among the group of the formula: Q1-X1- wherein X1 represents oxygen atom (O); Q1 represents aryl-(C1-C6)-alkyl, heteroaryl, heteroaryl-(C1-C6)-alkyl, heterocyclyl or heterocyclyl-(C1-C6)-alkyl and wherein neighboring carbon atoms in any (C2-C6)-alkylene chain in substitute at R1 are separated optionally by insertion to the chain the group taken among oxygen atom (O) and N(R5) wherein R5 represents hydrogen atom or (C1-C6)-alkyl, or when the inserted group represents N(R5); R5 can represent also (C2-C6)-alkanoyl and wherein any group -CH2 or -CH3 in substitute R1 carries one or more substitutes in each indicated group -CH2 or -CH3 and wherein these substitutes are taken among halogen atom or (C1-C6)-alkyl, or substitute taken among hydroxy-, amino-group, (C1-C6)-alkoxy-, (C1-C6)-alkylthio-group, (C1-C6)-alkylsulfinyl, (C1-C6)-alkylsulfonyl, (C1-C6)-alkylamino-, di-[(C1-C6)-alkyl]amino-, (C2-C6)-alkanoyloxy, (C2-C6)-alkanoylamino- and N-(C1-C6)-alktyl-(C2-C6)-alkanoylamino-group, or among the group of the formula: -X3-Q3wherein X3 represents oxygen atom (O) and Q3 represents heteroaryl, and wherein any aryl, heteroaryl or heterocyclyl group in substitute at R1 carries optionally 1, 2 or 3 substitutes that can be similar or different and taken among halogen atom, trifluoromethyl, cyano-, hydroxy-, amino-group, carbamoyl, (C1-C6)-alkyl, (C2-C8)-alkenyl, (C2-C8)-alkynyl, (C1-C6)-alkoxy-, (C2-C6)-alkenyloxy, (C2-C6)-alkynyloxy,(C1-C6)-alkylthio-group, (C1-C)-alkylsulfinyl, (C1-C6)-alkylsulfonyl, (C1-C6)-alkylamino-, di-[(C1-C6)-alkyl]amino-group, (C1-C6)-alkoxycarbonyl, N-(C1-C6)-alkylcarbamoyl, N,N-di-[(C1-C6)-alkyl]carbamoyl, (C2-C6)-alkanoyl, (C2-C6)-alkanoyloxy-, (C2-C)-alkanoylamino- and N-(C1-C6)-alkyl-(C2-C6)-alkanoylamino-group, or among the group of the formula: -X4-R8 wherein X4 represents a simple bond and R8 represents hydroxy-(C1-C6)-alkyl, (C1-C6)-alkoxy-(C1-C6)-alkyl, cyano-(C1-C6)-alkyl, amino-(C1-C6)-alkyl, (C1-C6)-alkylamino-(C1-C6)-alkyl or di-[(C1-C6)-alkyl]amino-(C1-C6)-alkyl, or among the group of the formula: -X5-Q4 wherein X5 represents a simple bond or -CO, and Q4 represents heterocyclyl or heterocyclyl-(C1-C6)-alkyl that carries optionally 1 or 2 substitutes that can be similar or different and taken among halogen atom, (C1-C6)-alkyl, (C2-C8)-alkenyl, (C2-C8)-alkynyl and (C1-C6)-alkoxy-group and wherein any heterocyclyl group in substitute at R1 carries optionally 1 or 2 oxo-substitutes, and wherein any aryl group in the group R1 represents phenyl; any heteroaryl group in the group R1 is taken among pyrrolyl, imidazolyl, triazolyl and pyridyl, and any heterocyclyl group in the group R1 is taken among oxyranyl, tetrahydrofuranyl, tetrahydropyranyl, pyrrolidinyl, morpholinyl, 1,1-dioxotetrahydro-1,4-thiazinyl, piperidinyl, homopiperidinyl, piperazinyl and homopiperazinyl; R2 represents hydrogen atom; n = 0, 1, 2 or 3; R3 represents halogen atom, trifluoromethyl, cyano-, hydroxy-group, (C1-C6)-alkyl, (C2-C8)-alkenyl, (C2-C8)-alkynyl or (C1-C6)-alkoxy-group, or its pharmaceutically acceptable salt. Also, invention relates to methods for preparing compounds of the formula (1) and to pharmaceutical composition based on thereof for using as an anti-tumor agent. Invention provides preparing new derivatives of quinazoline possessing an anti-tumor activity.
EFFECT: improved preparing method, valuable medicinal properties of compounds and pharmaceutical composition.
17 cl, 7 tbl, 7 ex
Authors
Dates
2006-01-10—Published
2001-08-15—Filed