FIELD: chemistry.
SUBSTANCE: invention relates to the compounds of the formula (I) 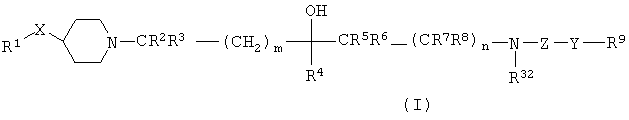 where: X is O; Y represents a bond, CH2, NR35, CH2NH, CH2NHC(O), CH(OH), CH(NHC(O)R33), CH(NHS(O)2R34), CH2O or CH2S; Z is C(O), or if Y is a bond, then Z can also be S(O)2; R1 could be substituted with phenyl; R4 is hydrogen, C1-6-alkyl (substituted possibly by C3-6-pilkoalkyl) or C3-6-cycloalkyl; R2, R3, R5, R6, R7 and R8 are independently hydrogen, C1-6-alkyl or C3-6-cycloalkyl; type independently indicate 0 or 1; R9 could possibly be substituted with an aryl or heterocycle; R10, R32 and R35 are independently hydrogen, C1-6-alkyl or C3-6-cycloalkyl; R33 and R34 are C1-6-alkyl or C3-6-cycloalkyl; where the aforesaid aryl and heterocyclic groups, when possible, can be substitute with: halogen cyanogens, nitro, hydroxyl, oxo, S(O)Kr12, OC(O)NR13R14, NR15R16, NR17C(O)R18, NR19C(O)NR20R21, S(O)2NR22R23, NR24S(O)2R25, C(O)NR26R27, C(O)R28, CO2R29, NR30CO2R31, by C1-6-alkyl (which itself can be monosubstituted with NHC(O)phenyl), C1-6-halogenalkyl, C1-6-alkoxy(C1-6)alkyl, C1-6-alkoxy, C1-6-halogenaloxy, C1-6-alkoxy(C1-6)-alkoxy, C1-6-alkylthio, C2-6-alkenyl, C2-6-alkinil, C3-10-cycloalkyl, methylenedioxy, difluoromethylenedioxy, phenyl, phenyl(C1-4)alkyl, phenoxy, phenylthio, phenyl(C1-4)alkyl, morpholinyl, heteroaryl, heteroaryl(C1-4)alkyl, heteroarylhydroxy of heteroaryl(C1-4)alkoxy, where any of the said phenyl and heteroaryl groups can be substituted by halogen, hydroxyl, nitro, S(O)r(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-.4-alkyl)2, cyanogens, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), CO2H, CO2(C1-4-alkyl), NHC(O)( C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)( C1-4-alkyl), CF3 or OCF3; k and r independently mean 0, 1 or 2; R13, R14, R15, R16, R17, R18, R19, R20, R21, R22, R23, R24, R26, R27, R29 and R30 independently represent hydrogen, C1-6-alkyl (probably replaced by halogen, hydroxyl or C3-10-cycloalkyl), CH2(C2-6-alkenyl), C3-6-cycloalkyl, phenyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), NH(C1-4-alkyl)2, S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2, cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), C(O)N(C1-4-alkyl)2, CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), CF3 or OCF3) or heterocyclyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), N(C1-4-alkyl)2, S(O)2)(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2, cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), C(O)N(C1-4-alkyl)2, CO2H5 CO2(C1-4-alkyl), NHC(O)( C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)( C1-4-alkyl), CF3 or OCF3); alternatively, NR13R14, NR15R16, NR20R21, NR22R23, NR26R27 can independently form 4-7-member heterocyclic ring, selected from the group, which includes: azetidine (which can be substituted by hydroxyl or C1-4-alkyl), pyrrolidine, piperidine, azepine, 1,4-morpholine or 1,4-piperazine, the latter is probably substituted by C1-4-alkyl on the peripheral nitrogen; R12, R25, R28 and R31 are independently C1-6-alkyl (possibly substituted by halogen, hydroxyl or C3-10-cycloalkyl), CH2(C2-6-alkenyl), phenyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4- alkyl), N(C1-4-alkyl)2, (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2 (and these alkyl groups can connec to form a ring as described hereabove for R13 and R14), cyanogen, C1-4- alkyl, C1-4- alkoxy, C(O)NH2, C(O)NH(C1-4- alkyl), C(O)N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), NHC(O)(C1-4-alkyl), CF3 or OCF3) or heterocyclyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), N(C1-4-alkyl)2, (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4- alkyl), C(O)N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), CF3 or OCF3); or its N-oxide; or its pharmaceutically acceptable salt, solvate or solvate of its salt, which are modulators of activity of chemokines (especially CCR3); also described is the pharmaceutical composition on their basis and the method of treating the chemokines mediated painful condition.
where: X is O; Y represents a bond, CH2, NR35, CH2NH, CH2NHC(O), CH(OH), CH(NHC(O)R33), CH(NHS(O)2R34), CH2O or CH2S; Z is C(O), or if Y is a bond, then Z can also be S(O)2; R1 could be substituted with phenyl; R4 is hydrogen, C1-6-alkyl (substituted possibly by C3-6-pilkoalkyl) or C3-6-cycloalkyl; R2, R3, R5, R6, R7 and R8 are independently hydrogen, C1-6-alkyl or C3-6-cycloalkyl; type independently indicate 0 or 1; R9 could possibly be substituted with an aryl or heterocycle; R10, R32 and R35 are independently hydrogen, C1-6-alkyl or C3-6-cycloalkyl; R33 and R34 are C1-6-alkyl or C3-6-cycloalkyl; where the aforesaid aryl and heterocyclic groups, when possible, can be substitute with: halogen cyanogens, nitro, hydroxyl, oxo, S(O)Kr12, OC(O)NR13R14, NR15R16, NR17C(O)R18, NR19C(O)NR20R21, S(O)2NR22R23, NR24S(O)2R25, C(O)NR26R27, C(O)R28, CO2R29, NR30CO2R31, by C1-6-alkyl (which itself can be monosubstituted with NHC(O)phenyl), C1-6-halogenalkyl, C1-6-alkoxy(C1-6)alkyl, C1-6-alkoxy, C1-6-halogenaloxy, C1-6-alkoxy(C1-6)-alkoxy, C1-6-alkylthio, C2-6-alkenyl, C2-6-alkinil, C3-10-cycloalkyl, methylenedioxy, difluoromethylenedioxy, phenyl, phenyl(C1-4)alkyl, phenoxy, phenylthio, phenyl(C1-4)alkyl, morpholinyl, heteroaryl, heteroaryl(C1-4)alkyl, heteroarylhydroxy of heteroaryl(C1-4)alkoxy, where any of the said phenyl and heteroaryl groups can be substituted by halogen, hydroxyl, nitro, S(O)r(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-.4-alkyl)2, cyanogens, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), CO2H, CO2(C1-4-alkyl), NHC(O)( C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)( C1-4-alkyl), CF3 or OCF3; k and r independently mean 0, 1 or 2; R13, R14, R15, R16, R17, R18, R19, R20, R21, R22, R23, R24, R26, R27, R29 and R30 independently represent hydrogen, C1-6-alkyl (probably replaced by halogen, hydroxyl or C3-10-cycloalkyl), CH2(C2-6-alkenyl), C3-6-cycloalkyl, phenyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), NH(C1-4-alkyl)2, S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2, cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), C(O)N(C1-4-alkyl)2, CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), CF3 or OCF3) or heterocyclyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), N(C1-4-alkyl)2, S(O)2)(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2, cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4-alkyl), C(O)N(C1-4-alkyl)2, CO2H5 CO2(C1-4-alkyl), NHC(O)( C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)( C1-4-alkyl), CF3 or OCF3); alternatively, NR13R14, NR15R16, NR20R21, NR22R23, NR26R27 can independently form 4-7-member heterocyclic ring, selected from the group, which includes: azetidine (which can be substituted by hydroxyl or C1-4-alkyl), pyrrolidine, piperidine, azepine, 1,4-morpholine or 1,4-piperazine, the latter is probably substituted by C1-4-alkyl on the peripheral nitrogen; R12, R25, R28 and R31 are independently C1-6-alkyl (possibly substituted by halogen, hydroxyl or C3-10-cycloalkyl), CH2(C2-6-alkenyl), phenyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4- alkyl), N(C1-4-alkyl)2, (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2 (and these alkyl groups can connec to form a ring as described hereabove for R13 and R14), cyanogen, C1-4- alkyl, C1-4- alkoxy, C(O)NH2, C(O)NH(C1-4- alkyl), C(O)N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), NHC(O)(C1-4-alkyl), CF3 or OCF3) or heterocyclyl (itself probably replaced by halogen, hydroxyl, nitro, NH2, NH(C1-4-alkyl), N(C1-4-alkyl)2, (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), S(O)2(C1-4-alkyl), S(O)2NH2, S(O)2NH(C1-4-alkyl), S(O)2N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), cyanogen, C1-4-alkyl, C1-4-alkoxy, C(O)NH2, C(O)NH(C1-4- alkyl), C(O)N(C1-4-alkyl)2 (and these alkyl groups can connect to form a ring as described hereabove for R13 and R14), CO2H, CO2(C1-4-alkyl), NHC(O)(C1-4-alkyl), NHS(O)2(C1-4-alkyl), C(O)(C1-4-alkyl), CF3 or OCF3); or its N-oxide; or its pharmaceutically acceptable salt, solvate or solvate of its salt, which are modulators of activity of chemokines (especially CCR3); also described is the pharmaceutical composition on their basis and the method of treating the chemokines mediated painful condition.
EFFECT: obtaining new compounds possessing useful biological properties.
13 cl, 238 ex
| Title | Year | Author | Number |
|---|---|---|---|
| COMPOUND AIMED AT PROTEIN AND DEGRADATION THEREOF, AND METHOD FOR PRODUCTION AND USE THEREOF | 2021 |
|
RU2829459C1 |
| CHIRAL DIARYL MACROCYCLES AS MODULATORS OF PROTEIN KINASES | 2016 |
|
RU2732405C2 |
| PIPERIDINE DERIVATIVES AS MODULATORS OF ACTIVITY OF CCR5 RECEPTOR, METHOD AND INTERMEDIATE COMPOUNDS FOR OBTAINING THEM, AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | 2002 |
|
RU2345990C2 |
| NEW PHTHALAZINONE DERIVATIVES AS AURORA A KINASE INHIBITORS | 2005 |
|
RU2397166C2 |
| COMPOUNDS AND METHODS FOR KINASE MODULATION AND INDICATIONS FOR THEIR USE | 2013 |
|
RU2666146C2 |
| NEW PHOSPHORUS-CONTAINING THYMOMIMETIC DRUGS | 2004 |
|
RU2422450C2 |
| SUBSTITUTED BETA-CARBOLINES, METHOD FOR THEIR PREPARING | 2001 |
|
RU2277095C2 |
| HETEROCYCLIC COMPOUNDS AND USES THEREOF | 2014 |
|
RU2680100C9 |
| CORTICOTROPIN-RELEASING FACTOR ANTAGONISTS | 1995 |
|
RU2221799C2 |
| CARBOCYCLIC AND HETEROCYCLIC ARYLSULFONES AS γ-SECRETASE INHIBITORS | 2007 |
|
RU2448964C2 |
Authors
Dates
2008-07-27—Published
2003-02-17—Filed