FIELD: chemistry.
SUBSTANCE: invention relates to novel methods of producing L-methionine, D-methionine or any mixture of L- and D-methionine from homoserine and is characterised by that L-homoserine, D-homoserine or mixtures of L- and D-homoserine of formula (I) 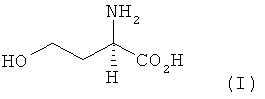 are chemically converted to methionine without formation of intermediate N-carbamoyl homoserine, 4-(2-bromoethyl)hydantoin and 4-(2-methylthioethyl)hydantoin. According to the first of the disclosed methods, chemical conversion involves a first step for ring closure in the presence of an acid catalyst to obtain the corresponding 2-amino-4-butyrolactone of formula III
are chemically converted to methionine without formation of intermediate N-carbamoyl homoserine, 4-(2-bromoethyl)hydantoin and 4-(2-methylthioethyl)hydantoin. According to the first of the disclosed methods, chemical conversion involves a first step for ring closure in the presence of an acid catalyst to obtain the corresponding 2-amino-4-butyrolactone of formula III 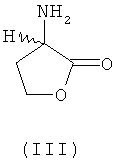 or salt thereof of formula IV
or salt thereof of formula IV 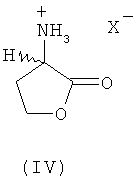 (values of X are given in claim 1), and at the second step that lactone or salt thereof, by reacting with MeSH, is converted to L-methionine, D-methionine or a mixture of L- and D-methionine. According to the second of the disclosed methods, chemical conversion consists of steps a) to c). At step a) homoserine of formula (I) undergoes basic catalytic N-acylation with an acylating agent and ring closure to N-acyl-L- and/or D-homoserine lactone of formula (VIII)
(values of X are given in claim 1), and at the second step that lactone or salt thereof, by reacting with MeSH, is converted to L-methionine, D-methionine or a mixture of L- and D-methionine. According to the second of the disclosed methods, chemical conversion consists of steps a) to c). At step a) homoserine of formula (I) undergoes basic catalytic N-acylation with an acylating agent and ring closure to N-acyl-L- and/or D-homoserine lactone of formula (VIII) 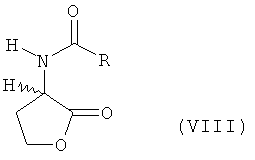 at temperature ranging from 20 to 100°C, preferably from 50 to 90°C, the acylating agent having general formula R-CO-X1 (values of R and X1 are given in claim 6). At step b) the N-acylhomoserine lactone obtained at step a), by reacting with MeSH in the presence of a base catalyst, is converted to the corresponding N-acylmethionine of formula (VII)
at temperature ranging from 20 to 100°C, preferably from 50 to 90°C, the acylating agent having general formula R-CO-X1 (values of R and X1 are given in claim 6). At step b) the N-acylhomoserine lactone obtained at step a), by reacting with MeSH in the presence of a base catalyst, is converted to the corresponding N-acylmethionine of formula (VII) 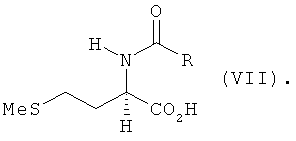 At step c) the N-actylmethionine obtained at step b) is hydrolysed at temperature higher than 95°C to the corresponding methionine. The base catalyst used at step b) is a trialkylamine of general formula NR3R4R5 (values of R3, R4 and R5 are given in claim 6) or DABCO, DBU, TBD, hexamethylenetetramine, tetramethylethylenediamine or tetramethyl guanidine. Step b) uses 1-20 mole equivalents, preferably 1-10 mole equivalents, of the base with respect to the hydroxide equivalent.
At step c) the N-actylmethionine obtained at step b) is hydrolysed at temperature higher than 95°C to the corresponding methionine. The base catalyst used at step b) is a trialkylamine of general formula NR3R4R5 (values of R3, R4 and R5 are given in claim 6) or DABCO, DBU, TBD, hexamethylenetetramine, tetramethylethylenediamine or tetramethyl guanidine. Step b) uses 1-20 mole equivalents, preferably 1-10 mole equivalents, of the base with respect to the hydroxide equivalent.
EFFECT: improved method.
18 cl, 1 tbl, 10 ex
| Title | Year | Author | Number |
|---|---|---|---|
| METHOD OF OBTAINING KETOACIDS AND THEIR DERIVATIVES | 2008 |
|
RU2536046C2 |
| 2-METHYLTHIOETHYL SUBSTITUTED HETEROCYCLES AS ADDITIVES TO FORAGE | 2008 |
|
RU2516833C2 |
| METHOD OF OBTAINING METHYLMERCAPTANE FROM DIALKYL SULPHIDES AND DIALKYL POLYSULPHIDES | 2008 |
|
RU2490255C2 |
| KETOMETHIONINE KETALS AND DERIVATIVES THEREOF | 2007 |
|
RU2483062C2 |
| SYNTHESIS AND USE OF 2-OXO-4-METHYLTHIOBUTANOIC ACID, SALTS AND DERIVATIVES THEREOF | 2005 |
|
RU2385862C2 |
| METHOD OF PRODUCING ANGIOTENSIN RECEPTOR BLOCKER | 2003 |
|
RU2412173C2 |
| OBTAINING AND USING METHIONYL-METHIONINE AS AN ADDITIVE TO FODDERS FOR FISHES AND CRUSTACEANS | 2009 |
|
RU2599783C2 |
| FODDER ADDITIVES REPRESENTED BY DIPEPTIDES | 2010 |
|
RU2536467C2 |
| PROCESS OF L-GLUFOSINATE AND L-GLUFOSINATE INTERMEDIATE PRODUCTION | 2019 |
|
RU2799336C2 |
| DERIVATIVES OF PIPERAZINE, METHOD OF THEIR SYNTHESIS AND PHARMACEUTICAL COMPOSITION | 1998 |
|
RU2194048C2 |
Authors
Dates
2013-01-20—Published
2007-01-04—Filed