FIELD: medicine, pharmaceutics.
SUBSTANCE: invention refers to substituted quinoxaline-type piperidine compounds of formula 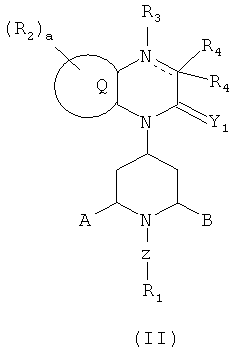 or to a pharmaceutically acceptable derivative thereof, wherein: Y1 represents O; Q is specified in condensed benzo or pyridino; each R2 is independently specified in: (a) -halogen or -CN; (b) -(C1-C6)alkyl; a is an integer specified in 0, 1 or 2; a dash line in a 6-member ring containing a nitrogen atom which is condensed with Q group means the presence or absence of a bond, and when the dash line means the absence of the bond, then R3, and one R4 are absent; R3 is specified in: (a) -H; each R4 is independently specified in: (a) -H; or (b) - halogen or CN; or (c) -X, -(C1-C6)alkyl-X, -(5- or 6-member)heterocyclyl-X or -(5- or 6-member)heterocyclyl-(C1-C6)alkyl-X; or (d) -C(=Y)X, -C(=Y)T3, -C(=Y)YX, - C(=Y)YT3, -C(-Y)N(T1)(T2), -C(=Y)N(R9)CN, -C(=Y)N(R9)X, -C(=Y)N(R9)YH, -C(=Y)N(R9)YX, -C(=Y)N(R9)YCH2X, -C(-Y)N(R9)YCH2CH2X or -C(=Y)N(R9)S(K))2T3; or (e) -N(R9)X, -N(R9)-CH2X, -N(R9)-CH2CH2X, -N(R9)CH2N(R9)C(=N(R12))N(R12)2, -N(R9)-CH2CH2N(R9)C(=N(RI2))N(R12)2, -N(T1)(T2), -N(T3)C(=Y)T3, -N(T3)C(=Y)YT3, -N(T3)C(=Y)N(T1)(T2), -N(T3)S(=O)2T3 or -N(T3)S(=O)2N(T1)(T2); X represents: (a) -H, -( C1-C6)alkyl, -(C2-C6)alkenyl, -(C1-C6)alkoxy, -(C3-C7)cycloalkyl, -(5- or 6-member)heterocycle or -(7-10-member)bicycloheterocycle each of which is unsubstituted or substituted with 1, 2 or 3 of optionally substituted R8 groups; or (b) -phenyl, -naphthalenyl, or -(5- or 6-member)heteroaryl each of which is unsubstituted or substituted with 1 or 2 of independently specified in R7 groups; each Y is independently specified in O; A and B are independently specified in: (a) -H; or (c) A-B together can form a (C2-C6)bridge each can optionally contain -HC=CH- or -O- in a (C2-C6)bridge; wherein the 6-member ring containing a nitrogen atom which is condensed with Q group can be found in the endo- or exo- configuration in relation to the A-B bridge; or (d) A-B together can form the -CH2-N(Ra)-CH2- bridge wherein the 6-member ring containing a nitrogen atom is condensed with Q group, and can be found in the endo- or exo- configuration in relation to the A-B bridge; Ra is specified in -H or -(C1-C6)alkyl; Z represents -[(C1-C10)alkyl optionally substituted with R1]h-, wherein h is equal to 0 or 1; each R1 is independently specified in: (b) -(C1-C10)alkyl, -(C2-C10)alkenyl, -(C2-C10)alkynyl3 -(C3-C7)cycloalkoxy, -(C6-C14)bicycloalkyl, -(C8-C10)tricycloalkyl, -(C5-C10)cycloalkenyl, -(C7-C14)bicycloalkenyl, -(3-7-member)heterocyclyl each of which is unsubtituted or substituted with 1, 2 or 3 of independently specified in R8 groups;
or to a pharmaceutically acceptable derivative thereof, wherein: Y1 represents O; Q is specified in condensed benzo or pyridino; each R2 is independently specified in: (a) -halogen or -CN; (b) -(C1-C6)alkyl; a is an integer specified in 0, 1 or 2; a dash line in a 6-member ring containing a nitrogen atom which is condensed with Q group means the presence or absence of a bond, and when the dash line means the absence of the bond, then R3, and one R4 are absent; R3 is specified in: (a) -H; each R4 is independently specified in: (a) -H; or (b) - halogen or CN; or (c) -X, -(C1-C6)alkyl-X, -(5- or 6-member)heterocyclyl-X or -(5- or 6-member)heterocyclyl-(C1-C6)alkyl-X; or (d) -C(=Y)X, -C(=Y)T3, -C(=Y)YX, - C(=Y)YT3, -C(-Y)N(T1)(T2), -C(=Y)N(R9)CN, -C(=Y)N(R9)X, -C(=Y)N(R9)YH, -C(=Y)N(R9)YX, -C(=Y)N(R9)YCH2X, -C(-Y)N(R9)YCH2CH2X or -C(=Y)N(R9)S(K))2T3; or (e) -N(R9)X, -N(R9)-CH2X, -N(R9)-CH2CH2X, -N(R9)CH2N(R9)C(=N(R12))N(R12)2, -N(R9)-CH2CH2N(R9)C(=N(RI2))N(R12)2, -N(T1)(T2), -N(T3)C(=Y)T3, -N(T3)C(=Y)YT3, -N(T3)C(=Y)N(T1)(T2), -N(T3)S(=O)2T3 or -N(T3)S(=O)2N(T1)(T2); X represents: (a) -H, -( C1-C6)alkyl, -(C2-C6)alkenyl, -(C1-C6)alkoxy, -(C3-C7)cycloalkyl, -(5- or 6-member)heterocycle or -(7-10-member)bicycloheterocycle each of which is unsubstituted or substituted with 1, 2 or 3 of optionally substituted R8 groups; or (b) -phenyl, -naphthalenyl, or -(5- or 6-member)heteroaryl each of which is unsubstituted or substituted with 1 or 2 of independently specified in R7 groups; each Y is independently specified in O; A and B are independently specified in: (a) -H; or (c) A-B together can form a (C2-C6)bridge each can optionally contain -HC=CH- or -O- in a (C2-C6)bridge; wherein the 6-member ring containing a nitrogen atom which is condensed with Q group can be found in the endo- or exo- configuration in relation to the A-B bridge; or (d) A-B together can form the -CH2-N(Ra)-CH2- bridge wherein the 6-member ring containing a nitrogen atom is condensed with Q group, and can be found in the endo- or exo- configuration in relation to the A-B bridge; Ra is specified in -H or -(C1-C6)alkyl; Z represents -[(C1-C10)alkyl optionally substituted with R1]h-, wherein h is equal to 0 or 1; each R1 is independently specified in: (b) -(C1-C10)alkyl, -(C2-C10)alkenyl, -(C2-C10)alkynyl3 -(C3-C7)cycloalkoxy, -(C6-C14)bicycloalkyl, -(C8-C10)tricycloalkyl, -(C5-C10)cycloalkenyl, -(C7-C14)bicycloalkenyl, -(3-7-member)heterocyclyl each of which is unsubtituted or substituted with 1, 2 or 3 of independently specified in R8 groups;

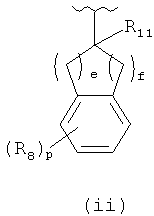 or
or 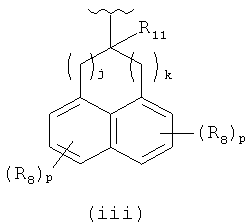 or (d) -phenyl, -naphthalenyl each of which is unsubstituted or substituted with R7 group; each R6 is optionally specified in -H; each R7 is independently specified in -(C1-C4)alkyl, -OR9, -C(halogen)3, -CH(halogen)2, -CH2(halogen), -CN, -halogen, -N(R9)2, -C(=O)OR9; each R8 is independently specified in -(C1-C4alkyl, tetrzolyl, imidazolyl, furanyl, -(C1-C6)alkylCOOR9, -OR9, -SR9, -C(halogen)3, -CH(halogen)2, -CH2(halogen), -CN, =O, -halogen, -N(R9)(C1-C6)alkylCOOR9, -N(R9)2, -N(R9)S(=O)2R12, -N(R9)C(=O)R12, -N(R9)C(=O)OR12, -C(=O)R9, -C(=O)N(T1)(T2), -C(=O)OR9, -OC(=O)R9, or -S(=O)2R9; each R9 is independently specified in -H, -(C1-C6)alkyl, -(C3-C8)cycloalkyl, -phenyl, -benzyl, -(5- to 6-member)heterocycle, -C(halogen)3; -CH(halogen)2 or -CH2(halogen); if h is equal to O, then R11 can be specified in -H, -C(=O)OR9 or -C(=O)N(R6)2 or R11 can be -(C1-C4)alkyl; if h is equal to 1, then R11 can be specified in -H; each R12 is independently specified in -H or -(C1-C4)alkyl; m is equal to an integer specified in 3, 4, 5, 6, 7, 8 or 9; each e and f is equal to an integer independently specified in 0 or 1, provided 2≤(e+f)≤5; each j and k is equal to an integer independently specified in 0 or 1, provided 1≤(j+k)≤4; each p is equal to an integer independently specified in 0 or 1; each T1, T2, and T3 is independently specified in -H or -(C1-C10)alkyl which is unsubstituted or substituted with 1, 2 or 3 from independently specified R8 groups, or T1 and T2 together can form 5- to 8-member ring wherein the number of ring atoms contains a nitrogen atom wherein T1 and T2 are bound; the above 5- to 8-member ring is unsubstituted or substituted with 1, 2 or 3 from independently specified R8 groups and optionally any carbon atom in the above 5- to 8-member ring is independently substituted with O or N(R6); each halogen is independently specified in -F, -CI, -Br or -I.
or (d) -phenyl, -naphthalenyl each of which is unsubstituted or substituted with R7 group; each R6 is optionally specified in -H; each R7 is independently specified in -(C1-C4)alkyl, -OR9, -C(halogen)3, -CH(halogen)2, -CH2(halogen), -CN, -halogen, -N(R9)2, -C(=O)OR9; each R8 is independently specified in -(C1-C4alkyl, tetrzolyl, imidazolyl, furanyl, -(C1-C6)alkylCOOR9, -OR9, -SR9, -C(halogen)3, -CH(halogen)2, -CH2(halogen), -CN, =O, -halogen, -N(R9)(C1-C6)alkylCOOR9, -N(R9)2, -N(R9)S(=O)2R12, -N(R9)C(=O)R12, -N(R9)C(=O)OR12, -C(=O)R9, -C(=O)N(T1)(T2), -C(=O)OR9, -OC(=O)R9, or -S(=O)2R9; each R9 is independently specified in -H, -(C1-C6)alkyl, -(C3-C8)cycloalkyl, -phenyl, -benzyl, -(5- to 6-member)heterocycle, -C(halogen)3; -CH(halogen)2 or -CH2(halogen); if h is equal to O, then R11 can be specified in -H, -C(=O)OR9 or -C(=O)N(R6)2 or R11 can be -(C1-C4)alkyl; if h is equal to 1, then R11 can be specified in -H; each R12 is independently specified in -H or -(C1-C4)alkyl; m is equal to an integer specified in 3, 4, 5, 6, 7, 8 or 9; each e and f is equal to an integer independently specified in 0 or 1, provided 2≤(e+f)≤5; each j and k is equal to an integer independently specified in 0 or 1, provided 1≤(j+k)≤4; each p is equal to an integer independently specified in 0 or 1; each T1, T2, and T3 is independently specified in -H or -(C1-C10)alkyl which is unsubstituted or substituted with 1, 2 or 3 from independently specified R8 groups, or T1 and T2 together can form 5- to 8-member ring wherein the number of ring atoms contains a nitrogen atom wherein T1 and T2 are bound; the above 5- to 8-member ring is unsubstituted or substituted with 1, 2 or 3 from independently specified R8 groups and optionally any carbon atom in the above 5- to 8-member ring is independently substituted with O or N(R6); each halogen is independently specified in -F, -CI, -Br or -I.
EFFECT: invention refers to the intermediate compounds of formula
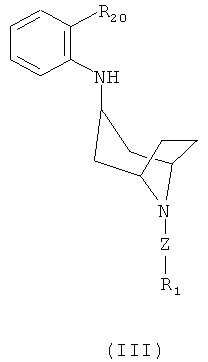 ,
, 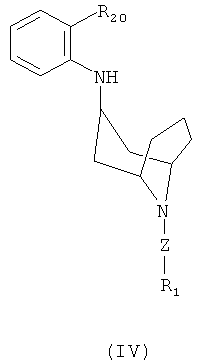 ,
, 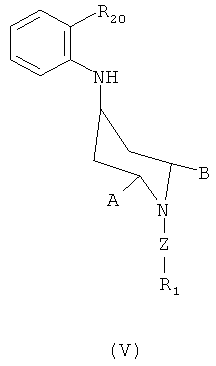 for preparing the above compounds of formula (II), compositions containing the above compounds and to a method of treating or preventing a diseased state, such as a pain.
for preparing the above compounds of formula (II), compositions containing the above compounds and to a method of treating or preventing a diseased state, such as a pain.
36 cl, 58 ex, 2 tbl
| Title | Year | Author | Number |
|---|---|---|---|
| TRPV1 ANTAGONISTS AND USE THEREOF | 2008 |
|
RU2452733C2 |
| 3,4-DIHYDROISOQUINOLINE COMPOUND AND USE THEREOF | 2021 |
|
RU2825312C1 |
| ANTIVIRAL COMPOUNDS | 2009 |
|
RU2505540C2 |
| SUBSTITUTED IMIDAZOPYRIDINYL-AMINO-PYRIDINE COMPOUNDS, USEFUL FOR TREATMENT OF CANCER | 2010 |
|
RU2619463C2 |
| DERIVATIVES OF GLYCOPEPTIDE OR THEIR SALTS, METHOD OF SYNTHESIS, PHARMACEUTICAL COMPOSITION | 1995 |
|
RU2145609C1 |
| NEUROACTIVE COMPOUNDS AND THEIR APPLICATION METHODS | 2015 |
|
RU2764702C2 |
| MONOMER DERIVATIVES OF GLYCOPEPTIDE ANTIBIOTIC | 2005 |
|
RU2424248C2 |
| TREATMENT OF ADDICTION AND IMPULSE-CONTROL DISORDERS USING PDE7 INHIBITORS | 2011 |
|
RU2661410C2 |
| ANTIVIRAL COMPOUNDS | 2009 |
|
RU2505539C2 |
| PIPERADINYL-SUBSTITUTED ISOQUINOLONE DERIVATIVES AS Rho-KINASE INHIBITORS | 2006 |
|
RU2414467C2 |
Authors
Dates
2013-07-27—Published
2008-08-29—Filed