FIELD: pharmaceuticals.
SUBSTANCE: invention relates to crystalline form I of the compound represented by formula (I), 6-ethyl-4-(4-methoxy-4-methylpiperidin-1-yl)-2-oxo-1,2-dihydro-1,7-diazanaphthalene- 3-carbonitrile, where the crystalline form is characterized by an x-ray powder diffraction pattern containing characteristic peaks at values of 2θ 7.3 ± 0.2°, 13.6 ± 0.2°, 14.5 ± 0.2°, 18.0 ± 0.2°, 19.1 ± 0.2°, 22.0 ± 0.2° and 23.4 ± 0.2°, as determined using Cu-Kα radiation. Crystalline Form I of the invention is prepared by dissolving the compound of formula (I) in either methanol or ethanol, raising the temperature to reflux until completely dissolved, and cooling naturally to room temperature for crystallization; or by completely dissolving a compound of formula (I) in one solvent — dichloromethane or in a mixed solvent — dichloromethane/water/isopropanol, and evaporating one solvent or mixed solvent until the system is saturated and crystalline form I is precipitated. The invention also relates to a pharmaceutical preparation for treatment or prevention of the diseases mediated by PDE9, containing crystalline Form I according to the invention and one or more pharmaceutical carriers.
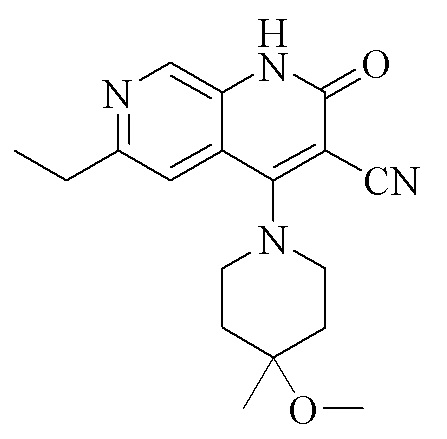 (I)
(I)
EFFECT: crystalline form of 6-ethyl-4-(4-methoxy-4-methylpiperidin-1-yl)-2-oxo-1,2-dihydro-1,7-diazanaphthalene-3-carbonitrile has excellent physical and chemical properties and pharmaceutical stability, as well as pharmacodynamic and pharmacokinetic properties.
6 cl, 18 dwg, 10 tbl, 36 ex
Authors
Dates
2024-02-29—Published
2020-03-13—Filed