FIELD: medicine.
SUBSTANCE: invention relates to therapy, and can be used to determine the presence of combined pathology—5 and more comorbidities in therapeutic patients. Patient’s sex, body mass index (BMI), blood pressure, low-density lipoprotein (LDL) cholesterol concentrations are determined, high-density lipoprotein (HDL) alpha-cholesterol, blood serum glucose. Concentration of C-reactive protein (CRP) in blood serum is determined, presence of non-alcoholic fatty liver disease (NAFLD), arterial hypertension (AH) is detected in the examined patient. According to the BMI index, the presence of obesity is detected, the LDL cholesterol content is used to detect the presence of low-density lipoprotein hypercholesterolemia (hyper LDL cholesterol), high-density lipoprotein hypocholesterolemia (HDL hypo-cholesterol) is detected by the content of alpha-cholesterol HDL. A calculated coefficient Y is determined for assessing the severity of the combined pathology by formula Y=3.387451+0.3994689*x1+1.240534*x2+0.6011361*x3+0.392105*x4+0.07857636*x5+0.0695682*x6+0.001535169*x7−1.427382*x8, where Y is the design coefficient; x1 is equal to 1 for male patients or 2 for female patients, x2 is equal to 0 in the absence of NAFLD or 1 in the presence of NAFLD, x3 is equal to 0 in the absence of AH or 1 in the presence of AH, x4 is 0 in the absence of obesity or 1 in the presence of obesity, x5 is equal to 0 in the absence of hyper LDL cholesterol or 1 in the presence thereof, x6 is equal to 0 in the absence of hypo HDL cholesterol or 1 in the presence thereof, x7 is the concentration blood serum glucose (mmol/l), x8—concentration of CRP in blood serum (mg/l), *—sign of multiplication. Presence of a combined pathology is determined by formula 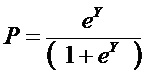 , where P is an indicator of the presence of a combined pathology; e is Euler's number = 2.7182. If the P value is 0.5 to 1.0, the presence of a combined pathology is determined. If P is from 0.0 to 0.49, the absence of combined pathology is determined.
, where P is an indicator of the presence of a combined pathology; e is Euler's number = 2.7182. If the P value is 0.5 to 1.0, the presence of a combined pathology is determined. If P is from 0.0 to 0.49, the absence of combined pathology is determined.
EFFECT: method provides the possibility of detecting at screening examination of patients with risk of high level of combined pathology corresponding to simultaneous presence of 5 and more nosologies, due to formation of group of indicators of patients' health.
1 cl, 8 tbl, 7 ex
| Title | Year | Author | Number |
|---|---|---|---|
| TRECRESAN APPLICATION FOR TREATING PATIENTS SUFFERING ARTERIAL HYPERTENSION AND OBESITY | 2010 |
|
RU2429832C1 |
| METHOD OF PREDICTING THE EFFECTIVENESS OF LITHOLYSIS IN CHOLELITHIASIS IN COMBINATION WITH NON-ALCOHOLIC FATTY LIVER DISEASE | 2023 |
|
RU2813033C1 |
| METHOD OF COMPLEX THERAPY OF OBESITY IN PATIENTS WITH ARTERIAL HYPERTENSION | 2015 |
|
RU2607186C1 |
| METHOD FOR PREDICTING CARDIOVASCULAR COMPLICATIONS IN PATIENTS WITH LOW OR MODERATE CARDIOVASCULAR RISK BY ASSESSING THEIR PSYCHOLOGICAL STATUS | 2020 |
|
RU2750716C1 |
| METHOD FOR PREDICTION OF THE RISK OF DEVELOPING ATHEROSCLEROTIC INVOLVEMENT OF CAROTID ARTERIES IN PATIENTS SUFFERING SYSTEMIC LUPUS ERYTHEMATOSUS | 2020 |
|
RU2736610C1 |
| METHOD OF INCREASING COMMITMENT TO HYPOLIPIDEMIC THERAPY | 2011 |
|
RU2470579C1 |
| METHOD FOR VERIFYING CHRONIC HEPATIC PATHOLOGY IN PATIENTS WITH ARTERIAL HYPERTENSION | 2013 |
|
RU2547246C1 |
| METHOD OF PATIENTS WITH OBESITY AND ARTERIAL HYPERTENSION STATE CORRECTION | 2019 |
|
RU2729352C1 |
| METHOD OF CONDITION CORRECTION IN PATIENTS WITH ARTERIAL HYPERTENSION AND OBESITY CORRECTION | 2019 |
|
RU2740374C2 |
| METHOD FOR ASSESSING ATHEROGENICITY OF APOLIPOPROTEIN B-CONTAINING LIPOPROTEINS | 2015 |
|
RU2601117C1 |
Authors
Dates
2024-11-19—Published
2024-03-06—Filed