FIELD: chemistry of organophosphorus compounds, biochemistry, medicine, pharmacy.
SUBSTANCE: invention relates to new bisamidate phosphonate compounds that are inhibitors of fructose 1,6-bis-phosphatase. Invention describes a compound of the formula (IA): 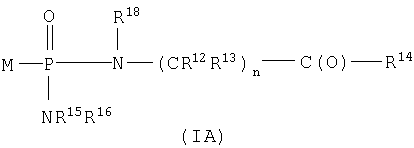 wherein compound of the formula (IA) is converted in vivo or in vitro to compound of the formula M-PO3H2 that is inhibitor of fructose 1,6-bis-phosphatase and wherein M represents R5-X- wherein R5 is chosen from a group consisting of compounds of the formula
wherein compound of the formula (IA) is converted in vivo or in vitro to compound of the formula M-PO3H2 that is inhibitor of fructose 1,6-bis-phosphatase and wherein M represents R5-X- wherein R5 is chosen from a group consisting of compounds of the formula 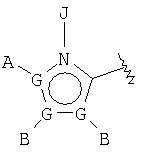 or
or 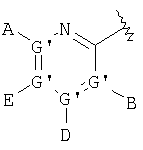 wherein each G is chosen from the group consisting of atoms C, N, O, S and Se and wherein only one G can mean atom O, S or Se and at most one G represents atom N; each G' is chosen independently from the group consisting of atoms C and N and wherein two G' groups, not above, represent atom N; A is chosen from the group consisting of -H, -NR4 2, -CONR4 2, -CO2R3, halide, -S(O)R3, -SO2R3, alkyl, alkenyl, alkynyl, perhaloidalkyl, haloidalkyl, aryl, -CH2OH, -CH2NR4 2, -CH2CN, -CN, -C(S)NH2, -OR2, -SR2, -N3, -NHC(S)NR4 2, -NHAc, or absent; each B and D is chosen independently from the group consisting of -H, alkyl, alkenyl, alkynyl, aryl, alicyclyl, aralkyl, alkoxyalkyl, -C(O)R11, -C(O)SR11, -SO2R11, -S(O)R3, -CN, -NR9 2, -OR3, -SR3, perhaloidalkyl, halide, -NO2, or absent and all groups except for -H, -CN, perhaloidalkyl, -NO2 and halide are substituted optionally; E is chosen from the group consisting of -H, alkyl, alkenyl, alkynyl, aryl, alicyclyl, alkoxyalkyl, -C(O)OR3, -CONR4 2, -CN, -NR9 2, -NO2, -OR3, -SR3, perhaloidalkyl, halide, or absent; all groups except for -H, -CN, perhaloidalkyl and halide are substituted optionally; J is chosen from the group consisting of -H, or absent; X represents optionally substituted binding group that binds R5 with phosphorus atom through 2-4 atoms comprising 0-1 heteroatom chosen from atoms N, O and S with exception that if X represents urea or carbamate then there are 2 heteroatoms that determine the shortest distance between R5 and phosphorus atom and wherein atom bound with phosphorus means carbon atom and wherein X is chosen from the group consisting of -alkyl(hydroxy)-, -alkynyl-, - heteroaryl-, -carbonylalkyl-, -1,1-dihaloidalkyl-, -alkoxyalkyl-, -alkyloxy-, -alkylthioalkyl-, -alkylthio-, -alkylaminocarbonyl-, -alkylcarbonylamino-, -alkoxycarbonyl-, -carbonyloxyalkyl-, -alkoxycarbonylamino- and -alkylaminocarbonylamino- and all groups are substituted optionally; under condition that X is not substituted with -COOR2, -SO3H or -PO3R2 2; n means a whole number from 1 to 3; R2 is taken among the group -R3 and -H; R3 is chosen from the group consisting of alkyl, aryl, alicyclyc and aralkyl; each R4 is chosen independently from the group consisting of -H and alkyl, or R4 and R4 form cycloalkyl group; each R9 is chosen independently from the group consisting of -H, alkyl, aryl, aralkyl and alicyclyl, or R9 and R9 form in common cycloalkyl group; R11 is chosen from the group consisting of alkyl, aryl, -NR2 2 and -OR2; each R12 and R13 is chosen independently from the group consisting of hydrogen atom (H), lower alkyl, lower aryl, lower aralkyl wherein all groups are substituted optionally, or R12 and R13 in common are bound through 2-5 atoms comprising optionally 1-2 heteroatoms chosen from the group consisting of atoms O, N and S to form cyclic group; each R14 is chosen independently from the group consisting of -OR17, -N(R17)2, -NHR17, -NR2OR19 and -SR17; R15 is chosen from the group consisting of -H, lower alkyl, lower aryl, lower aralkyl, or in common with R16 is bound through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; R16 is chosen from the group consisting of -(CR12R13)n-C(O)-R14, -H, lower alkyl, lower aryl, lower aralkyl, or in common with R15 is bound through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; each R17 is chosen independently from the group consisting of lower alkyl, lower aryl and lower aralkyl and all groups are substituted optionally, or R17 and R17 at atom N are bound in common through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; R18 is chosen independently among the group consisting of hydrogen atom (H), lower alkyl, aryl, aralkyl, or in common with R12 is bound through 1-4 carbon atoms forming cyclic group; each R19 is chosen independently from the group consisting of -H, lower alkyl, lower aryl, lower alicyclyl, lower aralkyl and -COR3; and under condition that when G' represents nitrogen atom (N) then the corresponding A, B, D or E are absent; at least one from A and B, or A, B, D and E is chosen from the group consisting of -H, or absent; when G represents nitrogen atom (N) then the corresponding A or B is not halide or group bound directly with G through a heteroatom; and its pharmaceutically acceptable salts. Also, invention describes a method for treatment or prophylaxis of diabetes mellitus, a method for inhibition of activity 0f fructose 1,6-bis-phosphatase, a method for decreasing blood glucose in animals, a method for treatment of diseases associated with glycogen deposition, a method for inhibition of gluconeogenesis in animal and a pharmaceutical composition based on compounds of the formula (IA).
wherein each G is chosen from the group consisting of atoms C, N, O, S and Se and wherein only one G can mean atom O, S or Se and at most one G represents atom N; each G' is chosen independently from the group consisting of atoms C and N and wherein two G' groups, not above, represent atom N; A is chosen from the group consisting of -H, -NR4 2, -CONR4 2, -CO2R3, halide, -S(O)R3, -SO2R3, alkyl, alkenyl, alkynyl, perhaloidalkyl, haloidalkyl, aryl, -CH2OH, -CH2NR4 2, -CH2CN, -CN, -C(S)NH2, -OR2, -SR2, -N3, -NHC(S)NR4 2, -NHAc, or absent; each B and D is chosen independently from the group consisting of -H, alkyl, alkenyl, alkynyl, aryl, alicyclyl, aralkyl, alkoxyalkyl, -C(O)R11, -C(O)SR11, -SO2R11, -S(O)R3, -CN, -NR9 2, -OR3, -SR3, perhaloidalkyl, halide, -NO2, or absent and all groups except for -H, -CN, perhaloidalkyl, -NO2 and halide are substituted optionally; E is chosen from the group consisting of -H, alkyl, alkenyl, alkynyl, aryl, alicyclyl, alkoxyalkyl, -C(O)OR3, -CONR4 2, -CN, -NR9 2, -NO2, -OR3, -SR3, perhaloidalkyl, halide, or absent; all groups except for -H, -CN, perhaloidalkyl and halide are substituted optionally; J is chosen from the group consisting of -H, or absent; X represents optionally substituted binding group that binds R5 with phosphorus atom through 2-4 atoms comprising 0-1 heteroatom chosen from atoms N, O and S with exception that if X represents urea or carbamate then there are 2 heteroatoms that determine the shortest distance between R5 and phosphorus atom and wherein atom bound with phosphorus means carbon atom and wherein X is chosen from the group consisting of -alkyl(hydroxy)-, -alkynyl-, - heteroaryl-, -carbonylalkyl-, -1,1-dihaloidalkyl-, -alkoxyalkyl-, -alkyloxy-, -alkylthioalkyl-, -alkylthio-, -alkylaminocarbonyl-, -alkylcarbonylamino-, -alkoxycarbonyl-, -carbonyloxyalkyl-, -alkoxycarbonylamino- and -alkylaminocarbonylamino- and all groups are substituted optionally; under condition that X is not substituted with -COOR2, -SO3H or -PO3R2 2; n means a whole number from 1 to 3; R2 is taken among the group -R3 and -H; R3 is chosen from the group consisting of alkyl, aryl, alicyclyc and aralkyl; each R4 is chosen independently from the group consisting of -H and alkyl, or R4 and R4 form cycloalkyl group; each R9 is chosen independently from the group consisting of -H, alkyl, aryl, aralkyl and alicyclyl, or R9 and R9 form in common cycloalkyl group; R11 is chosen from the group consisting of alkyl, aryl, -NR2 2 and -OR2; each R12 and R13 is chosen independently from the group consisting of hydrogen atom (H), lower alkyl, lower aryl, lower aralkyl wherein all groups are substituted optionally, or R12 and R13 in common are bound through 2-5 atoms comprising optionally 1-2 heteroatoms chosen from the group consisting of atoms O, N and S to form cyclic group; each R14 is chosen independently from the group consisting of -OR17, -N(R17)2, -NHR17, -NR2OR19 and -SR17; R15 is chosen from the group consisting of -H, lower alkyl, lower aryl, lower aralkyl, or in common with R16 is bound through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; R16 is chosen from the group consisting of -(CR12R13)n-C(O)-R14, -H, lower alkyl, lower aryl, lower aralkyl, or in common with R15 is bound through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; each R17 is chosen independently from the group consisting of lower alkyl, lower aryl and lower aralkyl and all groups are substituted optionally, or R17 and R17 at atom N are bound in common through 2-6 atoms comprising optionally 1 heteroatom chosen from the group consisting of atoms O, N and S; R18 is chosen independently among the group consisting of hydrogen atom (H), lower alkyl, aryl, aralkyl, or in common with R12 is bound through 1-4 carbon atoms forming cyclic group; each R19 is chosen independently from the group consisting of -H, lower alkyl, lower aryl, lower alicyclyl, lower aralkyl and -COR3; and under condition that when G' represents nitrogen atom (N) then the corresponding A, B, D or E are absent; at least one from A and B, or A, B, D and E is chosen from the group consisting of -H, or absent; when G represents nitrogen atom (N) then the corresponding A or B is not halide or group bound directly with G through a heteroatom; and its pharmaceutically acceptable salts. Also, invention describes a method for treatment or prophylaxis of diabetes mellitus, a method for inhibition of activity 0f fructose 1,6-bis-phosphatase, a method for decreasing blood glucose in animals, a method for treatment of diseases associated with glycogen deposition, a method for inhibition of gluconeogenesis in animal and a pharmaceutical composition based on compounds of the formula (IA).
EFFECT: valuable medicinal and biochemical properties of compounds.
69 cl, 7 tbl, 64 ex
Authors
Dates
2006-04-10—Published
2000-12-22—Filed