FIELD: chemistry.
SUBSTANCE: invention relates to inhibitors of leukotriene A4-hydrolase (LTA4H) of formula (II), their enatiomers, racemates and pharmaceutically acceptable salts, as well as a pharmaceutical composition based on said inhibitors and method of treating, preventing or suppressing inflammation and other conditions which are mediated by activity of leukotriene A4-hydrolase. In general formula (II) 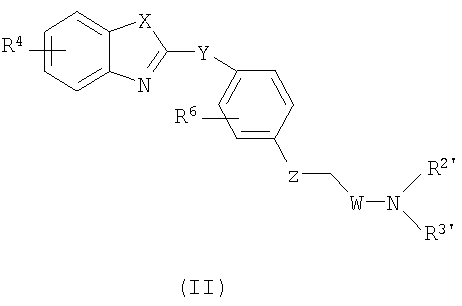 , X is chosen from a group which consists of NR5, O and S, where R5 is one of H and CH3; Y is O; Z is chosen from a group which consists of O and a bond; W is chosen from a group which consists of CH2 and CHR1-CH2, where R1 is H or OH, and where the carbon group bonded to R1 in the said CHR1-CH2 is not directly bonded to the nitrogen atom which is bonded to the said W; R4 is chosen from a group which consists of H, OCH3 and Cl; R6 is H or F; and R2' and R3' are each independently chosen from a group which consists of: A) H, C1-7alkyl, C3-7cycloalkyl, C3-7cycloalkyl-C1-7alkyl, where each of substitutes (A) is independently substituted with 0 or 1 RQ, where each of said RQ is a carbon atom substitute, which is at least one carbon atom, separate from nitrogen atom; B) HetRa substitute; C) -C1-7alkyl-C(O)Rx; H) -C0-4alkyl-Ar5, where Ar5 is a 5-member heteroaryl, which has one heteroatom, chosen from a group >NRY, and 0 or 1 additional heteroatom -N=, and optionally contains two carbonyl groups, and optionally benzo-condensed; I) -C0-4alkyl-Ar5' , where Ar5' is a 5-member heteroaryl, which contains 3 or 4 nitrogen atoms; M) SO2C1-4alkyl; alternatively, R2' and R3', taken together with a nitrogen atom with which they are bonded, form a heterocyclic ring which contains at least one heteroatom, which is the said bonded nitrogen atom, where the said heterocyclic ring is chosen from a group which consists of i) 4-7-member heterocyclic ring HetRb, where the said 4-7-member heterocyclic ring HetRb has one heteroatom, which is the said bonded nitrogen atom, and is substituted with 0, 1 or 2 identical or different substitutes, where the said substitutes are chosen from a group which consists of -RY, -CN, -C(O)RY, -C0-4alkyl-CO2RY, -C0-4alkyl-C(O)CO2RY, -C0-4alkyl-ORY, -C0-4alkyl-C(O)NRYRZ-, -C0-4alkyl-NRYC(O)RZ-, -C(O)NRZORY, -C0-4alkyl-NRYCO2RY, -C0-4alkyl-NRYC(O)NRYRY, -C0-4alkyl-NRYC(S)NRYRZ, -NRYC(O)CO2RY, -C0-4alkyl-NRWSO2RY, 1,3-dihydrobenzoimidazol-2-on-1-yl, 1-RY-1H-tetrazol-5-yl, RY-triazolyl, 2-RY-2H-tetrazol- 5-yl, -C0-4alkyl-C(O)N(RY)(SO2RY), -C0-4alkyl-N(RY)(SO2)NRYRY, -C0-4alkyl-N(RY)(SO2)NRYCO2RY, halogen,
, X is chosen from a group which consists of NR5, O and S, where R5 is one of H and CH3; Y is O; Z is chosen from a group which consists of O and a bond; W is chosen from a group which consists of CH2 and CHR1-CH2, where R1 is H or OH, and where the carbon group bonded to R1 in the said CHR1-CH2 is not directly bonded to the nitrogen atom which is bonded to the said W; R4 is chosen from a group which consists of H, OCH3 and Cl; R6 is H or F; and R2' and R3' are each independently chosen from a group which consists of: A) H, C1-7alkyl, C3-7cycloalkyl, C3-7cycloalkyl-C1-7alkyl, where each of substitutes (A) is independently substituted with 0 or 1 RQ, where each of said RQ is a carbon atom substitute, which is at least one carbon atom, separate from nitrogen atom; B) HetRa substitute; C) -C1-7alkyl-C(O)Rx; H) -C0-4alkyl-Ar5, where Ar5 is a 5-member heteroaryl, which has one heteroatom, chosen from a group >NRY, and 0 or 1 additional heteroatom -N=, and optionally contains two carbonyl groups, and optionally benzo-condensed; I) -C0-4alkyl-Ar5' , where Ar5' is a 5-member heteroaryl, which contains 3 or 4 nitrogen atoms; M) SO2C1-4alkyl; alternatively, R2' and R3', taken together with a nitrogen atom with which they are bonded, form a heterocyclic ring which contains at least one heteroatom, which is the said bonded nitrogen atom, where the said heterocyclic ring is chosen from a group which consists of i) 4-7-member heterocyclic ring HetRb, where the said 4-7-member heterocyclic ring HetRb has one heteroatom, which is the said bonded nitrogen atom, and is substituted with 0, 1 or 2 identical or different substitutes, where the said substitutes are chosen from a group which consists of -RY, -CN, -C(O)RY, -C0-4alkyl-CO2RY, -C0-4alkyl-C(O)CO2RY, -C0-4alkyl-ORY, -C0-4alkyl-C(O)NRYRZ-, -C0-4alkyl-NRYC(O)RZ-, -C(O)NRZORY, -C0-4alkyl-NRYCO2RY, -C0-4alkyl-NRYC(O)NRYRY, -C0-4alkyl-NRYC(S)NRYRZ, -NRYC(O)CO2RY, -C0-4alkyl-NRWSO2RY, 1,3-dihydrobenzoimidazol-2-on-1-yl, 1-RY-1H-tetrazol-5-yl, RY-triazolyl, 2-RY-2H-tetrazol- 5-yl, -C0-4alkyl-C(O)N(RY)(SO2RY), -C0-4alkyl-N(RY)(SO2)NRYRY, -C0-4alkyl-N(RY)(SO2)NRYCO2RY, halogen, 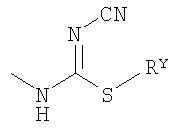 ,
, 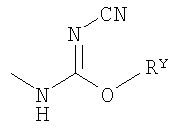 ,
,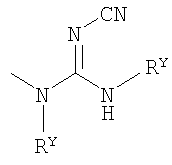 ; ii) 5-7-member heterocyclic ring HetRC which has one additional heteroatom separated from the said bonded nitrogen atom by at least one carbon atom, where the said additional heteroatom is chosen from a group which consists of O, S(=O)2 and >NRM, where the said 5-7-member heterocyclic ring HetRC has 0 or 1 carbonyl group and is substituted with 0, 1 or 2 substitutes at identical or different substituted carbon atoms, where the said substitutes are chosen from a group which consists of -C(O)RY and RZ; iii) one of 1H-tetrazol-1-yl, where 1H-tetrazol-1-yl is substituted at the carbon atom by 0 or 1 substitute such as -C0-4alkyl-RZ, -C0-4alkyl-CO2RY; and iv) one of benzimidazol-1-yl, 2,8-diazospiro[4.5]decan-1-on-8-yl, 4-{[(2-tert-butoxycarbonylaminocyclobutanecarbonyl)amino]methyl}piperidin-1-yl, 4-{[(2-aminocyclobutanecarbonyl)amino]methyl}piperidin-1-yl, 9-yl-tert-butyl ether 3,9-diazaspiro[5.5]undecane-3-carboxylic acid, 4-oxo-1-phenyl-1,3,8-triazaspiro[4.5]dec-8-yl, and where substitute HetRa is a 6-member heterocyclic ring, with a carbon atom at the bonding site and contains a >NRM group as a heteroatom, where the said heteroatom is separated from the said carbon atom at the bonding site with at least 1 additional carbon atom; Rk is chosen from a group which consists of H and -C1-4alkyl; RL is chosen from a group which consists of -CO2RS; RS is hydrogen; RM is chosen from a group which consists of RZ, -C(O)RY; RN is chosen from a group which consists of OCH3, CI, F, Br, I, OH, NH2, CN, CF3, CH3 and NO2; RQ is chosen from a group which consists of -CN, -C0-4alkyl-ORY, -C0-4alkyl-CO2RY, -C0-4alkyl-NRYRY, -C0-4alkyl-NRYCORY, -C0-4alkyl-NRYCONRYRZ, -C0-4alkyl-NRYSO2RY; RW is chosen from a group which consists of RY; RX is chosen from a group which consists of -ORY, -NRYRZ, -C1-4alkyl and -C1-4alkyl-RAr; RY is chosen from a group which consists of H, C1-4alkyl, -C0-4alkyl-RAr and -C0-4alkyl-RAr', each of which is substituted with 1 or 2 RN substitutes; RZ is chosen from a group which consists of RY, -C1-2alkyl-CO2RY ; RAr is a radical with a carbon atom at the bonding position, where the said radical is chosen from a group which consists of phenyl, pyridyl and pyrazinyl, where each carbon atom with permissible valence in each of the said groups is independently substituted with at least 0, 1 or 2 RN or 0 or 1 RL; RAr' is a 5-6-member ring which has 1 or 2 heteroatoms, chosen from a group which consists of O, S, N and >NRY, and has 0 or 2 unsaturated bonds and 0 or 1 carbonyl group, where each member with permissible valence in each of the said rings is independently substituted with 0 or 1 or 2 RK; Description is given of inhibitors of leukotriene A4-hydrolase (LTA4H) of formula (II), a composition which contains these inhibitions, and their use for inhibiting activity of the LTA4H enzyme, as well as for treating, preventing or suppressing inflammation and/or conditions which are associated with such inflammation. In the said formula (I): X is chosen from a group which consists of NR5, O and S, where R5 is one of H and CH3; Y is chosen from a group which consists of CH2 and O, W is chosen from a group which consists of CH2 and CHR1-CH2, where R1 is H or OH, and where the carbon group bonded to R1 in the said CHR1-CH2 is not directly bonded to a nitrogen atom; R4 is chosen from a group which consist of H, OCH3, CI, F, Br, OH, NH2, CN, CF3 and CH3; R6 is H or F; and R2 and R3 are each independently chosen from different groups.
; ii) 5-7-member heterocyclic ring HetRC which has one additional heteroatom separated from the said bonded nitrogen atom by at least one carbon atom, where the said additional heteroatom is chosen from a group which consists of O, S(=O)2 and >NRM, where the said 5-7-member heterocyclic ring HetRC has 0 or 1 carbonyl group and is substituted with 0, 1 or 2 substitutes at identical or different substituted carbon atoms, where the said substitutes are chosen from a group which consists of -C(O)RY and RZ; iii) one of 1H-tetrazol-1-yl, where 1H-tetrazol-1-yl is substituted at the carbon atom by 0 or 1 substitute such as -C0-4alkyl-RZ, -C0-4alkyl-CO2RY; and iv) one of benzimidazol-1-yl, 2,8-diazospiro[4.5]decan-1-on-8-yl, 4-{[(2-tert-butoxycarbonylaminocyclobutanecarbonyl)amino]methyl}piperidin-1-yl, 4-{[(2-aminocyclobutanecarbonyl)amino]methyl}piperidin-1-yl, 9-yl-tert-butyl ether 3,9-diazaspiro[5.5]undecane-3-carboxylic acid, 4-oxo-1-phenyl-1,3,8-triazaspiro[4.5]dec-8-yl, and where substitute HetRa is a 6-member heterocyclic ring, with a carbon atom at the bonding site and contains a >NRM group as a heteroatom, where the said heteroatom is separated from the said carbon atom at the bonding site with at least 1 additional carbon atom; Rk is chosen from a group which consists of H and -C1-4alkyl; RL is chosen from a group which consists of -CO2RS; RS is hydrogen; RM is chosen from a group which consists of RZ, -C(O)RY; RN is chosen from a group which consists of OCH3, CI, F, Br, I, OH, NH2, CN, CF3, CH3 and NO2; RQ is chosen from a group which consists of -CN, -C0-4alkyl-ORY, -C0-4alkyl-CO2RY, -C0-4alkyl-NRYRY, -C0-4alkyl-NRYCORY, -C0-4alkyl-NRYCONRYRZ, -C0-4alkyl-NRYSO2RY; RW is chosen from a group which consists of RY; RX is chosen from a group which consists of -ORY, -NRYRZ, -C1-4alkyl and -C1-4alkyl-RAr; RY is chosen from a group which consists of H, C1-4alkyl, -C0-4alkyl-RAr and -C0-4alkyl-RAr', each of which is substituted with 1 or 2 RN substitutes; RZ is chosen from a group which consists of RY, -C1-2alkyl-CO2RY ; RAr is a radical with a carbon atom at the bonding position, where the said radical is chosen from a group which consists of phenyl, pyridyl and pyrazinyl, where each carbon atom with permissible valence in each of the said groups is independently substituted with at least 0, 1 or 2 RN or 0 or 1 RL; RAr' is a 5-6-member ring which has 1 or 2 heteroatoms, chosen from a group which consists of O, S, N and >NRY, and has 0 or 2 unsaturated bonds and 0 or 1 carbonyl group, where each member with permissible valence in each of the said rings is independently substituted with 0 or 1 or 2 RK; Description is given of inhibitors of leukotriene A4-hydrolase (LTA4H) of formula (II), a composition which contains these inhibitions, and their use for inhibiting activity of the LTA4H enzyme, as well as for treating, preventing or suppressing inflammation and/or conditions which are associated with such inflammation. In the said formula (I): X is chosen from a group which consists of NR5, O and S, where R5 is one of H and CH3; Y is chosen from a group which consists of CH2 and O, W is chosen from a group which consists of CH2 and CHR1-CH2, where R1 is H or OH, and where the carbon group bonded to R1 in the said CHR1-CH2 is not directly bonded to a nitrogen atom; R4 is chosen from a group which consist of H, OCH3, CI, F, Br, OH, NH2, CN, CF3 and CH3; R6 is H or F; and R2 and R3 are each independently chosen from different groups.
EFFECT: new compounds have useful biological activity.
43 cl, 8 tbl, 12 dwg, 484 ex
| Title | Year | Author | Number |
|---|---|---|---|
| DERIVATIVES OF BENZIMIDAZOL, BENZOTIAZOL AND BENZOXAZOL AND THEIR APPLICATION AS LTA4H MODULATORS | 2004 |
|
RU2359970C2 |
| BENZOIMIDAZOLES AS PROLYL HYDROXYLASE INHIBITORS | 2009 |
|
RU2531354C2 |
| GLYCOLATOXIDASE INHIBITORS FOR THE TREATMENT OF DISEASES | 2018 |
|
RU2805308C2 |
| HETEROCYCLIC COMPOUNDS AND METHODS OF APPLICATION | 2009 |
|
RU2525116C2 |
| NOVEL FUSED PYRIMIDINE DERIVATIVES FOR INHIBITION OF TYROSINE KINASE ACTIVITY | 2011 |
|
RU2585177C2 |
| BROMDOMAIN INHIBITORS | 2012 |
|
RU2671571C1 |
| BROMDOMAIN INHIBITORS | 2012 |
|
RU2647592C2 |
| QUINAZOLINONES AS PROLYL HYDROXYLASE INHIBITORS | 2010 |
|
RU2528412C2 |
| 2,5-DISUBSTITUTED 3-MERCAPTOPENTANIC ACIDS AND METHOD OF OBTAINING THEM | 2003 |
|
RU2365583C2 |
| AZAADAMANTANE DERIVATIVES AND METHODS FOR APPLYING | 2007 |
|
RU2450002C2 |
Authors
Dates
2009-11-20—Published
2004-07-27—Filed