FIELD: chemistry.
SUBSTANCE: invention relates to uridine diphosphate-3-O-(hydroxymyristoyl)-N-acetylglucosamine deacetylase (LpxC) enzyme inhibitors of formula (I) 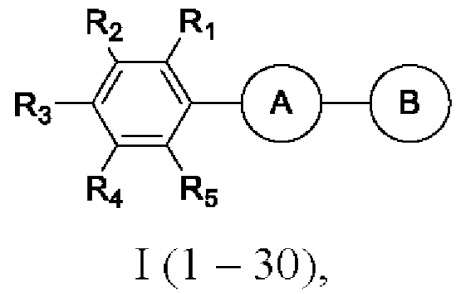 where A is 3,5-disubstituted nucleus of 1,2,4-oxadiazole (in the form of two regioisomers 1 and 2) or 2,5-disubstituted nucleus of 1,3,4-oxadiazole 3:
where A is 3,5-disubstituted nucleus of 1,2,4-oxadiazole (in the form of two regioisomers 1 and 2) or 2,5-disubstituted nucleus of 1,3,4-oxadiazole 3: 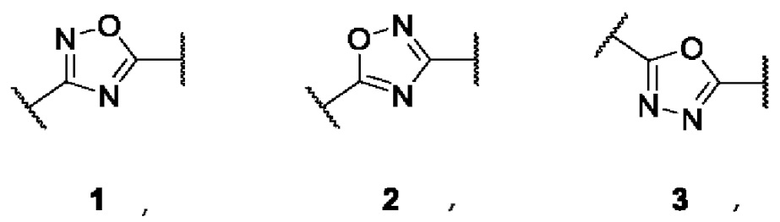 B is a functional group of hydroxamic acid (C(O)NHOH), free or conjugated with an aliphatic or aromatic linker:
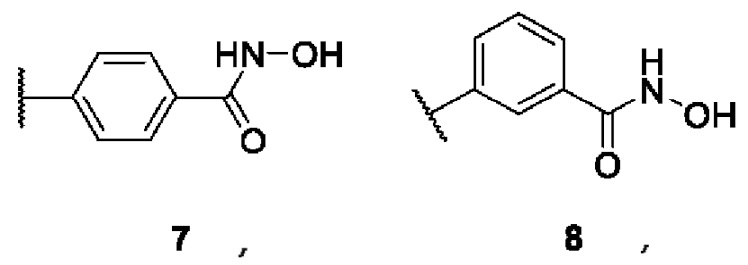
B is a functional group of hydroxamic acid (C(O)NHOH), free or conjugated with an aliphatic or aromatic linker: 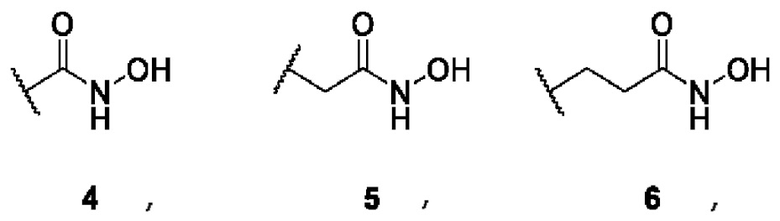
 R1 is H, F, H3CO, H3C; R2 is H, F, C1, I, H3CO, H3C; R3 is H, F, C1, I, H3CO, F3CO; R4 is H, F; R5 is H, F.
R1 is H, F, H3CO, H3C; R2 is H, F, C1, I, H3CO, H3C; R3 is H, F, C1, I, H3CO, F3CO; R4 is H, F; R5 is H, F.
EFFECT: synthesis of new chemical compounds, which are inhibitors of the LpxC enzyme, having pharmacological activity, a decrease in the cytotoxicity of the synthesized chemical compounds, which are inhibitors of the LpxC enzyme, improvement of ADME-properties of synthesized chemical compounds, which are inhibitors of LpxC enzyme, is shown.
35 cl, 11 tbl, 99 ex, 13 dwg
| Title | Year | Author | Number |
|---|---|---|---|
| METHOD FOR INHIBITING TUMOR CELLS WITH NEW DERIVATIVES OF 3-TRIFLUOROMETHYLQUINOXALINE 1,4-DIOXIDE | 2020 |
|
RU2746395C1 |
| DERIVATIVES OF 1,4-DIOXIDE OF QUINOXALINE-2-CARBONITRILE INHIBITING TUMOUR CELLS GROWTH | 2016 |
|
RU2640304C1 |
| QUATERNARY AMMONIUM SALTS BASED ON DERIVATIVES OF VITAMIN B6 | 2015 |
|
RU2607522C1 |
| 2-((4-(4-(BENZYLOXY)PYRIMIDIN-2-YL)PIPERAZIN-1-YL)METHYL)-1H-BENZO[d]IMIDAZOLE DERIVATIVES AS LOW-MOLECULAR GLUCAGON-LIKE PEPTIDE-1 RECEPTOR AGONISTS | 2024 |
|
RU2838323C1 |
| PYRIMIDINE DERIVATIVES AS KINASE INHIBITORS AND THERAPEUTIC APPLICATIONS THEREOF | 2015 |
|
RU2701188C2 |
| INHIBITORS OF FORMATION OF GLYCATION END PRODUCTS BASED ON AZO-DERIVATIVE PHENYLSULPHONIC ACIDS | 2016 |
|
RU2634594C1 |
| 8,10-DINITRO-DEHYDRO-1,5-5H,11H-[1,2,3,4]TETRAZINO[5',6':4,5][1,2,3]TRIAZOLO[2,1-a][1,2,3]BENZOTRIASEOL-1,3-DIOXIDE AND METHOD OF ITS PRODUCTION | 2017 |
|
RU2643363C1 |
| AZO-DERIVATIVES OF AMINOPHENOLS, CAPABLE OF INHIBITING FORMATION OF ADVANCED GLYCATION END PRODUCTS | 2024 |
|
RU2839138C1 |
| SUBSTITUTED PHENYLNAPHTHALENES AS ESTROGENIC AGENTS | 2002 |
|
RU2314283C2 |
| PYRIDOXINE-BASED COMPOUNDS CAPABLE OF GLUCOKINASE ENZYME ACTIVATION | 2017 |
|
RU2644355C1 |
Authors
Dates
2024-11-18—Published
2023-12-08—Filed