FIELD: medicine; pharmaceutics.
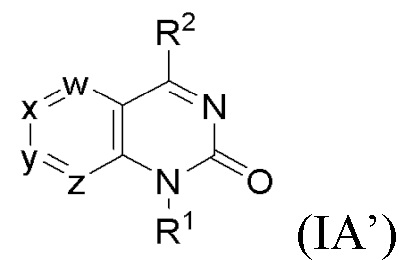
SUBSTANCE: invention relates to a compound of formula (IA') or a pharmaceutically acceptable salt thereof, which have inhibitory action on methionine adenosyltransferase 2A (MAT2A). In formula (IA') w is CR3 or N; x is CR4 or N; y is CR5 or N; z is CR6 or N, provided that not more than two of w, x, y and z are N, R3 is a hydrogen atom, C1-6 alkyl, C1-6 alkoxy, halogen, C1-6 haloalkyl, C1-6 haloalkoxy, C3-6 cycloalkyl, C3-6 cycloalkyl-C1-6 alkyloxy, cyano, C1-6 hydroxyalkoxy, C1-6 aminoalkoxy, heteroaryl, heterocyclyl or heterocyclyloxy, where heterocyclyl or heteroaryl separately or as part of another group is unsubstituted or is substituted with Ra, Rb and/or Rc independently selected from C1-6 alkyl, halogen and C1-6 alkoxycarbonyl; R5 is C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C1-6 alkoxy, halogen, C1-6 haloalkyl, C1-6 haloalkoxy, C3-6 cycloalkyl, cyano, C1-6 dialkylamino, C1-6 alkoxycarbonyl, C1-6 hydroxyalkyl, heteroaryl or heterocyclyl; R4 and R6 independently represent a hydrogen atom, C1-6 alkyl, C1-6 alkoxy, C1-6 alkylthio, halogen, C1-6 haloalkyl, C1-6 haloalkoxy or cyano; R1 is R7, where R7 is C3-6 cycloalkyl, C6-10 aryl or heteroaryl, where C6-10 aryl or heteroaryl is unsubstituted or substituted with Rd, Re and/or Rf; R2 is C1-6 alkyl, -O-R8, -NR9R10 or -Xb-R11, where R8 is C1-6 alkyl or C3-6 cycloalkyl; R9 is a hydrogen atom, C1-6 alkyl, C1-6 deuteroalkyl or C3-6 cycloalkyl; R10 is a hydrogen atom, C1-6 alkyl, C1-6 deuteroalkyl, C1-6 haloalkyl, C1-6 hydroxyalkyl, C1-6 alkoxy-C1-6 alkyl, C1-6 haloalkoxy-C1-6 alkyl, C1-6 aminoalkyl, C1-6 aminosulphonyl, C1-6 thioureidoalkyl, C1-6 alkylsulphonyl, C1-6 alkylsulfonyl-C1-6 alkyl, C1-6 cyanoalkyl, C1-6 alkylcarbonyl, C1-6 alkylaminocarbonyl, C1-6 dialkylaminocarbonyl, aminocarbonyl-C1-6 alkyl, C1-6 alkylaminocarbonyl-C1-6 alkyl, C1-6 dialkylaminocarbonyl-C1-6 alkyl, C3-6 cycloalkyl, C3-6 cycloalkyl-C1-6 alkyl, substituted C3-6 cycloalkyl, substituted C3-6 cycloalkyl-C1-6 alkyl, bridged cycloalkyl, C6-10 spirocycloalkyl, C6-10 aryl-C1-6 alkyl, heteroaryl, heteroaryl-C1-6 alkyl, heterocyclyl, heterocyclyl-C1-6 alkyl or spiroheterocyclyl, where aryl, heteroaryl or heterocyclyl separately or as part of another group is unsubstituted or substituted Rj, Rk and/or Rl; Xb is a bond or C1-6 alkylene; R11 is monocyclic heteroaryl or heterocyclyl selected from oxetanyl, azetidinyl, 2-oxo-azetidinyl, pyrrolidinyl, 2-oxo-pyrrolidinyl, piperidinyl and morpholinyl, where heteroaryl or heterocyclyl is unsubstituted or substituted Rm, Rn and/or Ro; Rd, Re, Rj, Rk, Rm and Rn are independently selected from C1-6 alkyl, C1-6 haloalkyl, C1-6 haloalkoxy, C1-6 alkoxy, hydroxy, C1-6 alkylsulphonyl, halogen, cyano, carboxy, C1-6 alkoxycarbonyl, C1-6 hydroxyalkyl, C1-6 alkoxy-C1-6 alkyl, and so forth; Rf, Rl and Ro are independently selected from C1-6 alkyl, C1-6 haloalkyl, C1-6 alkoxy, C1-6 haloalkoxy, C3-6 cycloalkyl, hydroxy, halogen, cyano, amino, C1-6 alkylamino, C3-6 cycloalkylsulfonylamino, carboxy-C1-6 alkyl and -Xc-R12, where Xc is a bond or C1-6 alkylene, R12 is unsubstituted C6-10 aryl. Each heteroaryl has from 5 to 10 ring members and from 1 to 3 heteroatoms in ring vertices, each independently selected from N, O or S; each heterocyclyl has 4 to 8 ring members and 1 to 2 heteroatoms at ring vertices, each independently selected from N, O or S(O)n, where n is 0, 1 or 2; each bridged cycloalkyl has from 5 to 7 members in a cycle in which two non-neighboring ring atoms are linked by a group (CRR’)q, where q is 1–3 and each R and R' independently represents H or methyl; and each spiroheterocyclyl is a saturated bicyclic ring having from 6 to 10 ring members and from 1 to 3 heteroatomic ring vertices, each independently selected from N, O or S(O)n, where n is 0, 1 or 2; and the bicyclic ring is bonded only through one atom. Invention also relates to a specific compound and a pharmaceutically acceptable salt thereof, a pharmaceutical composition containing said compounds, and methods of treating cancer.
EFFECT: there are disclosed 2-oxoquinazoline derivatives as methionine adenosyltransferase 2a inhibitors.
26 cl, 4 tbl, 16 ex
| Title | Year | Author | Number |
|---|---|---|---|
| 2,3-DIHYDROQUINAZOLINE COMPOUNDS AS NAV1.8 INHIBITORS | 2020 |
|
RU2833870C2 |
| HEXACYCLIC COMPOUNDS, PHARMACEUTICAL COMPOSITION CONTAINING THEREOF AND THEIR USING AS ANTITUMOR AGENTS, METHOD FOR PREPARING HEXACYCLIC COMPOUNDS | 2002 |
|
RU2299212C2 |
| SUBSTITUTED PYRROLES ACTIVE AS KINASES INHIBITORS | 2013 |
|
RU2666538C2 |
| PYRIMIDIN-4(3H)-ONE DERIVATIVES AS TRPV4 ANTAGONISTS | 2021 |
|
RU2840769C1 |
| AMINOPYRIMIDINE DERIVATIVES AND USE THEREOF AS AROMATIC HYDROCARBON RECEPTOR MODULATORS | 2021 |
|
RU2826628C1 |
| ARYLAMIDE DERIVATIVES AS TTX-S BLOCKERS | 2011 |
|
RU2535671C1 |
| PYRROLOPYRIMIDINES AS CFTR POTENTIATORS | 2017 |
|
RU2757457C2 |
| QUINOLINE DERIVATIVES AND MELK INHIBITORS CONTAINING SAME | 2011 |
|
RU2582610C2 |
| 1,2,4-TRIAZINE-3-AMINE DERIVATIVES | 2011 |
|
RU2771819C2 |
| CYCLIC IMINOPYRIMIDINE DERIVATIVES AS KINASE INHIBITORS | 2018 |
|
RU2801302C2 |
Authors
Dates
2024-11-14—Published
2019-12-09—Filed